 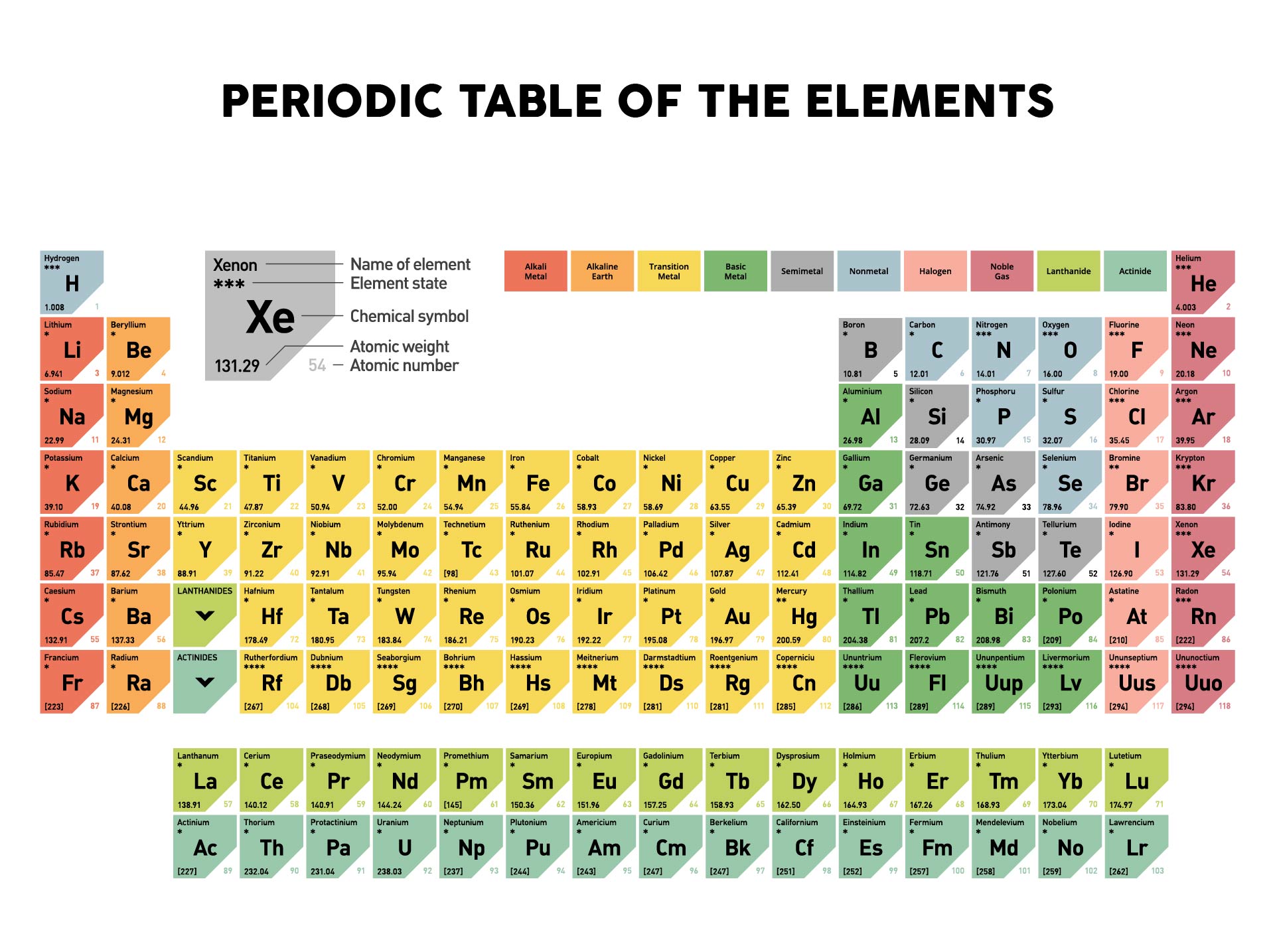
You will need to refer to a periodic table for proton values. From the periodic table: molar mass of hydrogen is 1 molar mass of oxygen is 16 molar mass of a water molecule 2(1) + 16 18 gm Now, to convert the gm into amu, all you have to do is multiply the gm you got by Avogadros number as follows: mass of water molecule 18 x 6.22 x 1023 1. In my calculations, I typically type the entire molar mass of each element at round at the very end and am able to get the same answer. It is better to round to the number of significant figures at the very end of your calculation. 1: Moles to Mass Conversion with Elements. In my discussion, we discussed that we will be expected to use significant figures when giving answers on the exam. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. One mole of Al atoms has a mass in grams that is numerically equivalent to the atomic mass of aluminum. In this notation, the atomic number is not included. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. 
1 The molar mass is a bulk, not molecular, property of a substance. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (C 12 H 22 O 11 (sucrose)). 
Then, lookup atomic weights for each element in periodic table: C: 12.0107, H: 1.00794, O: 15.9994. Visualize trends, 3D orbitals, isotopes, and mix compounds. First, compute the number of each atom in C 12 H 22 O 11 (sucrose): C: 12, H: 22, O: 11. Symbol-mass format for the above atom would be written as Cr-52. In chemistry, the molar mass ( M) of a chemical compound is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of said compound. Interactive periodic table showing names, electrons, and oxidation states. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. Use this visual tool for calculating molar mass for any chemical formula. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
